Czech ‘vaccine tours’ branded as FRAUD by Russian health ministry
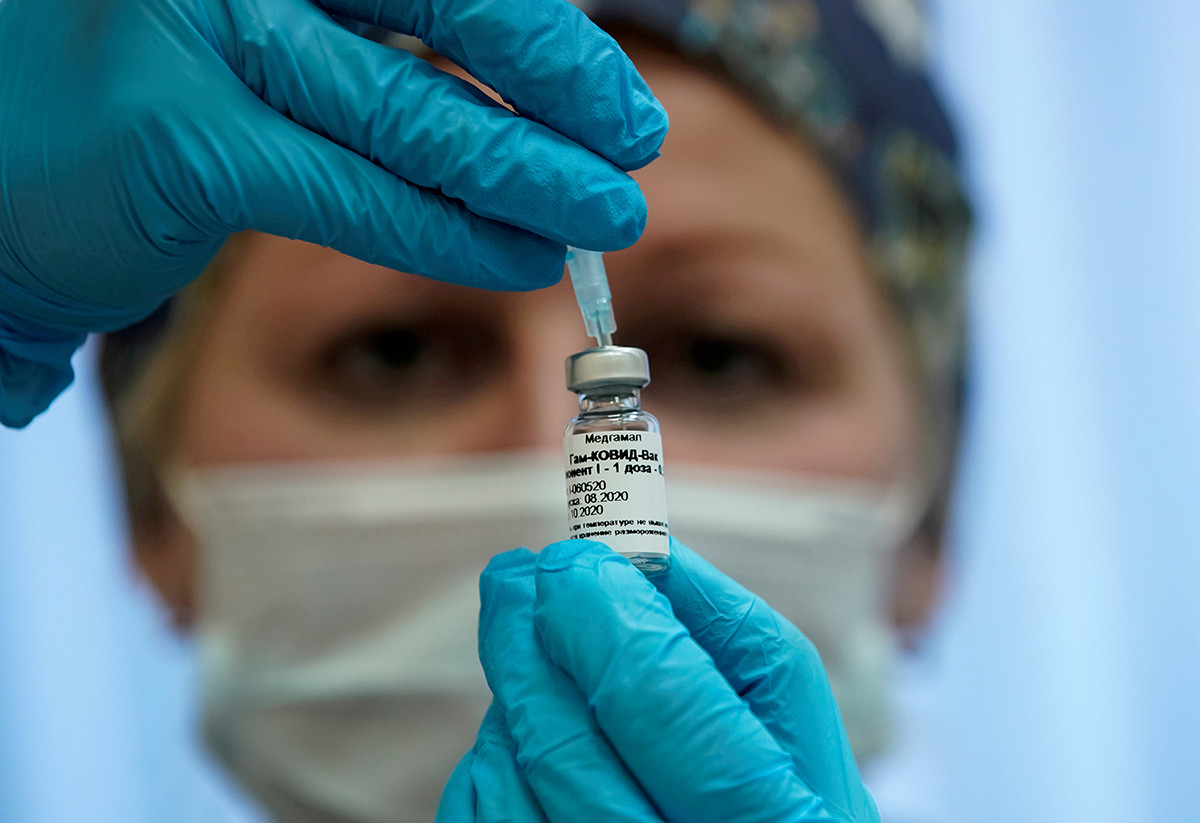
A nurse prepares Russia's "Sputnik-V" vaccine against the coronavirus disease (COVID-19) for inoculation in a post-registration trials stage at a clinic in Moscow
ReutersAndrey Konstantinov, a Prague-based entrepreneur of Russian descent, has told Czech newspaper ‘Hospodarske Noviny’ about his plans to organize tours to Moscow for Europeans, which would involve getting vaccinated for COVID-19. Czech media dubbed the idea “Sputnik trip”, in honor of the Russian vaccine’s name ‘Sputnik-V’, which became the first to be registered worldwide.
Booking the tour will cost 3,000 krona (approx. 10,000 rubles or $130), with the total price rising up to 30,000 krona. The proposed plan for the tour is as follows: The tourist obtains a Russian visa with the aid of an invitation provided on medical grounds, passes a COVID-19 test and purchases a ticket to Moscow.
On arrival in Russia, the visitor receives their first Sputnik-V injection (there are two). They can then stay in Moscow for a period of three weeks, before a second injection is administered, leading to the formation of antibodies in the patient’s organism.
Vaccine against COVID-19, made in Russia
Cadu Rolim/Keystone Press AgencyOnce that happens, the patient can leave. If desired, they can choose not to partake in the tour’s entertainment program between shots and return to the Czech Republic before the second injection. No information has been made available about the fun activities, or where visitors would stay during their stay.
The vaccine shots are scheduled to be administered at Moscow’s outpatient hospital ‘AO Medicina’ and will start running in November 2020, after the third phase of clinical trials for the Russian vaccine has completed, Konstantinov told Business FM radio.
“When the third phase completes, people will begin to travel. It’s a partnership agreement for the foreseeable future, meaning the next month [November]. It depends on how they solve those issues over there. We must provide some guarantees to people,” he added.
Konstantinov also spoke with ‘Kommersant’ newspaper, saying that work has begun on the waiting list, with several hundred names already on it.
The president of AO Medicina, Grigoriy Roytberg of the Russian Academy of Sciences, says that the outpatient hospital has no affiliation with the tours, nor is it running clinical trials on the Sputnik-V vaccine.
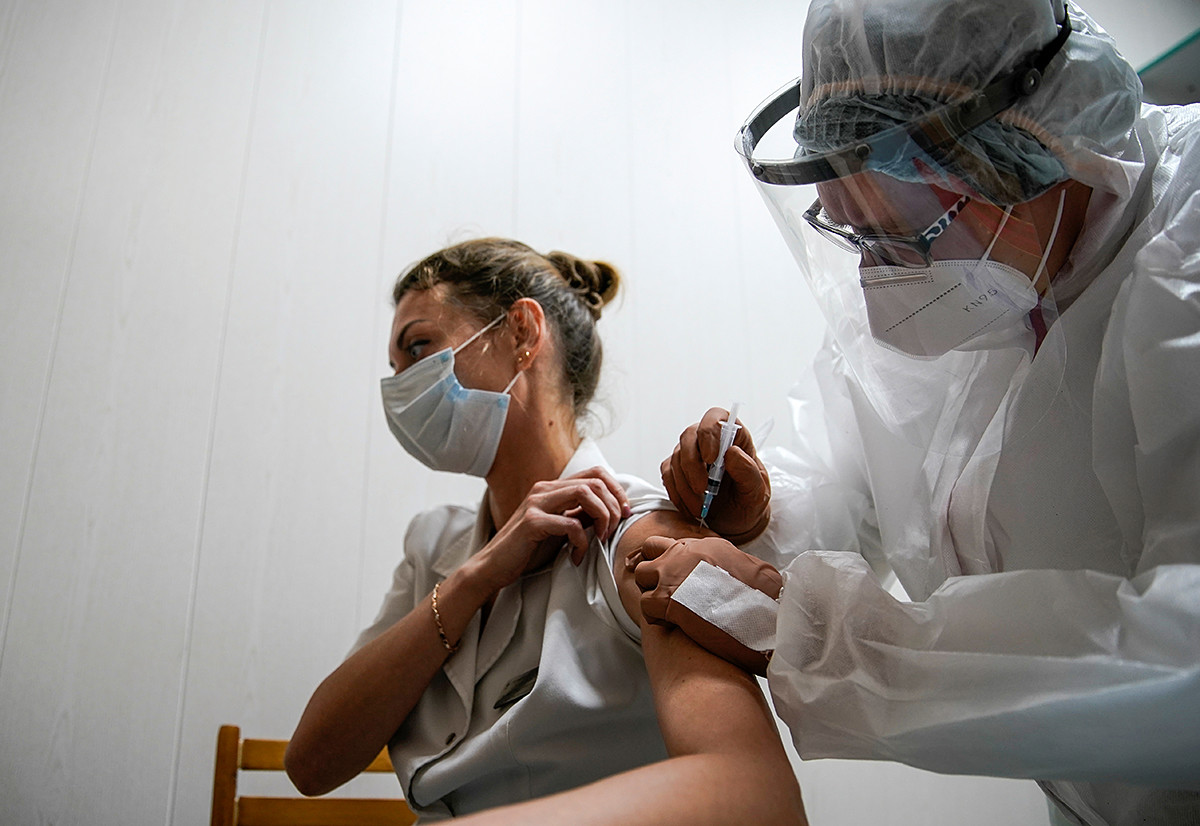
A medic of the regional hospital receives Russia's "Sputnik-V" vaccine shot against the coronavirus disease (COVID-19)
Reuters“It’s the first I hear of it, we do not do anything like that here. I don’t have these vaccines… We do medical tourism here, but it concerns completely different areas: radionuclide therapy, some oncological procedures - we get patients from across the world. But there was never any talk of COVID-19… There have never been any plans for vaccine tourism, nor are we planning [to do anything],” Roytberg told Business FM.
Commercial vaccination in Russia is still not possible, according to deputy minister of health Aleksey Kuznetsov.
“This is garden-variety fraud. The COVID-19 vaccine being offered commercially - let alone to foreign tourists, is impossible. The vaccination itself is already underway and is offered at no cost to Russian citizens. The absolute priority here are Russians,” Kuznetsov emphasized, adding that only once vaccination has been carried out for Russia’s at-risk population and once any other willing Rusisans receive the vaccine “can there be a discussion of any commercial deliveries”.
READ MORE: What we know about the FIRST registered COVID-19 vaccine in the world
If using any of Russia Beyond's content, partly or in full, always provide an active hyperlink to the original material.
Subscribe
to our newsletter!
Get the week's best stories straight to your inbox